
The IF7 lewis structure consists of iodine surrounded by 7 fluorine atoms. It is a structure with anomalies as illustrated in this article. 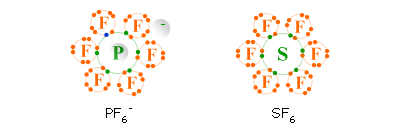
Sulfur hexafluoride (also a gas) consists of SF 6 molecules.IF7 lewis structure is an interhalogen compound which implies that halogen atoms are involved. Instead of an octet the phosphorus atom has 10 electrons in its valence shell. The dot diagram shows that P has a total of ten electrons on its valence. P is connected to two equally spaced F atoms below it and three equally spaced F atoms above it. 
Since each bond corresponds to a shared pair of electrons, the Lewis structure is It consists of PF 5 molecules in which each fluorine atom is bonded to the phosphorus atom. Phosphorus pentafluoride is a gas at room temperature. The overall charge of the molecule is negative 2.īCl 3 reacts with NH 3 in the following way:Įxamples of molecules with more than an octet of electrons are phosphorus pentafluoride (PF 5) and sulfur hexafluoride (SF 6). Thus BeCl 2 reacts with Cl – ions to form BeCl 4 – Ī B e C l 2 molecule reacts with two chlorine anions to give B e C l 4 with a complete octet set. Electron-deficient molecules typically react with species containing lone pairs, acquiring octets by formation of coordinate covalent bonds. Molecules such as BeCl 2 and BCl 3 are referred to as electron deficient because some atoms do not have complete octets. In which the valence shell of boron has only three pairs of electrons. The three pairs of unpaired electrons on chlorine are shown as dots. The three chlorine atoms are 60 degrees from one another and form a triangular shape with boron in the center. We are forced to write its structure asĪ central B atom is single bonded to three chlorine atoms. Similar arguments can be applied to boron trichloride, BCl 3, which is a stable gas at room temperature. Instead of an octet the valence shell of Be contains only two electron pairs. The three pairs of unpaired electrons on each chlorine is shown as dots. The structure isĪ central Be structure is single bonded to two chlorine atom. Since Cl atoms do not readily form multiple bonds, we expect the Be atom to be joined to each Cl atom by a single bond. Solid BeCl 2 has a relatively Complex structure at room temperature, but when it is heated to 750☌, a vapor which consists of separate BeCl 2 molecules is obtained. Beryllium dichloride, BeCl 2, is a covalent rather than an ionic substance. Good examples of the first type of exception are provided by BeCl 2 and BCl 3. Although elements such as Si, P, S, Cl, Br, and I obey the octet rule in many cases, under other circumstances they form more bonds than the rule allows. Elements in the third period and below can accommodate more than an octet of electrons.This usually occurs in compounds containing Be or B. Some stable molecules simply do not have enough electrons to achieve octets around all atoms.The octet rule is much less useful in dealing with compounds of the transition elements or inner transition elements, most of which involve some participation of d or f orbitals in bonding.Įven among the representative elements there are some exceptions to the Lewis theory. Therefore it is most successful in accounting for formulas of compounds of the representative elements, whose distinguishing electrons are also s and p electrons. Lewis’ theory concentrates on resemblances to noble-gas ns 2 np 6 valence octets. \)Ĭonsidering the tremendous variety in properties of elements and compounds in the periodic system, it is asking a great deal to expect a rule as simple as Lewis’ octet theory to be able to predict all formulas or to account for all molecular structures involving covalent bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
